To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Resource Library
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
A collection of videos showcased at our AAN 2026 Medical Affairs Booth.
Efgartigimod MoA, Indication Agnostic Video
Empasiprubart ARGX-117 Proposed Mechanism of Action video
Downloadable resources with key insights and clinical trial updates.

VYVGART/VYVGART HYTRULO MG and CIDP Cross Indication Safety Neurology Congress Handout
Overview of neurology-specific safety data for efgartigimod (including SC formulation) from global clinical trials

VYVGART gMG ADAPT NXT Manuscript Clinical Data Summary Sheet
summary of ADAPT NXT Part A clinical data

Immunology Innovation Program Flyer
Flyer highlighting the Immunology Innovation Program and commitment to co-creating novel antibody therapies

argenx Medical Affairs: Science of Collaboration Congress Handout
Double-gate folded handout for use at medical booths and symposia (with congress-specific disclaimers), first distributed at the ANA 2025 Trainee Breakfast for neurology trainees.

Cross-indication Externally Sponsored Research Handout
A snapshot of how argenx advances immunology innovation through deep scientific collaboration and antibody engineering.

Efgartigimod Mechanism of Action Handout
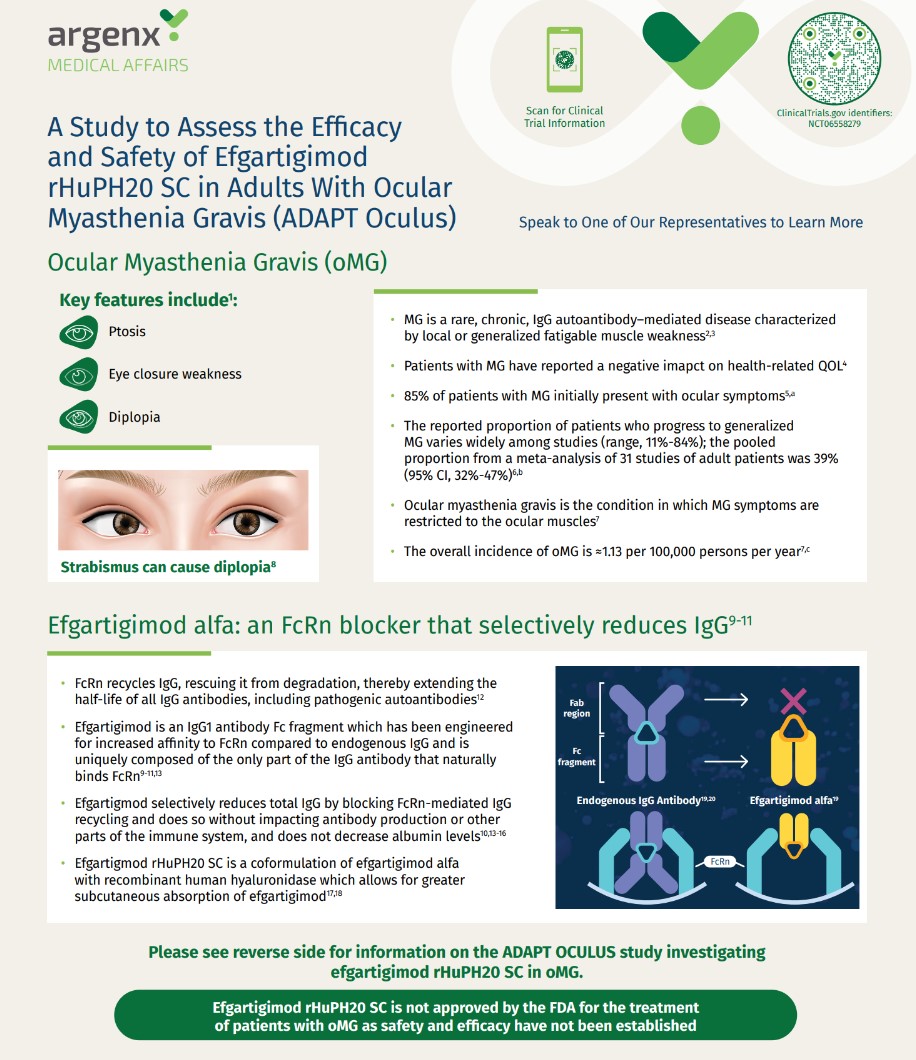
ADAPT Oculus Study Design Handout
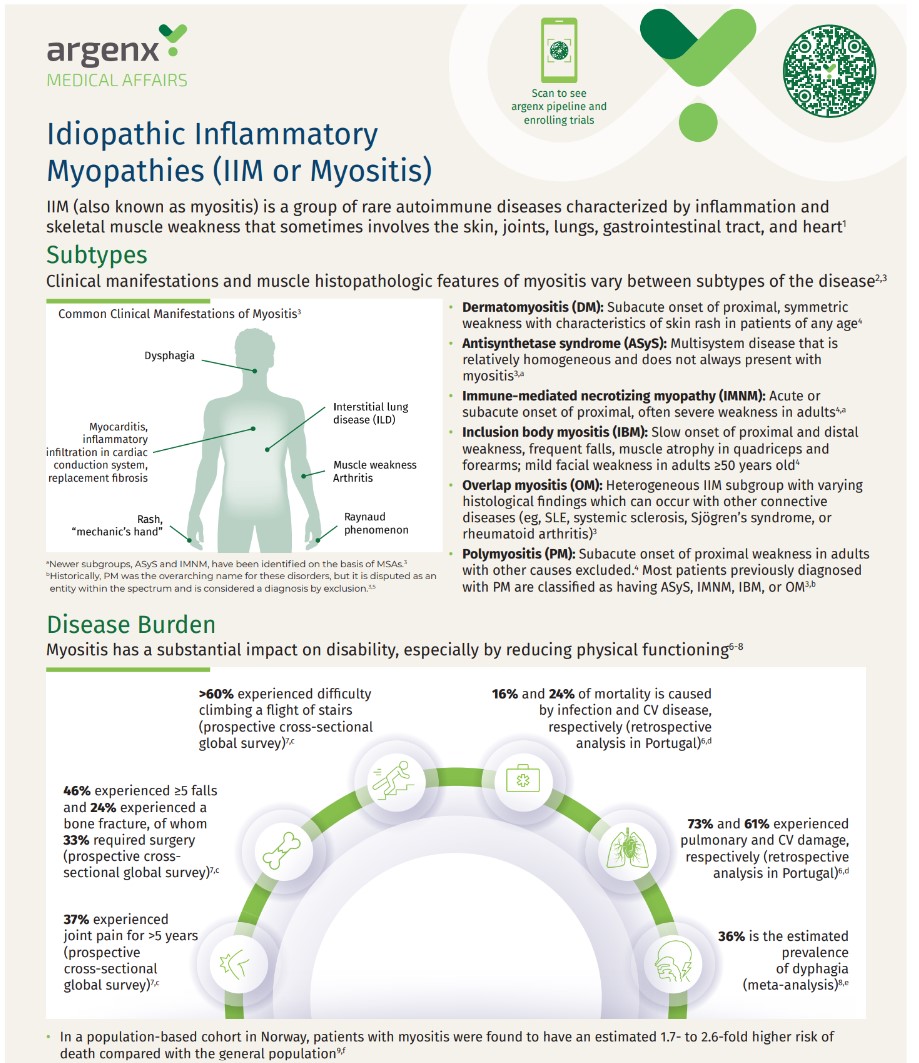
Myositis Disease State Handout
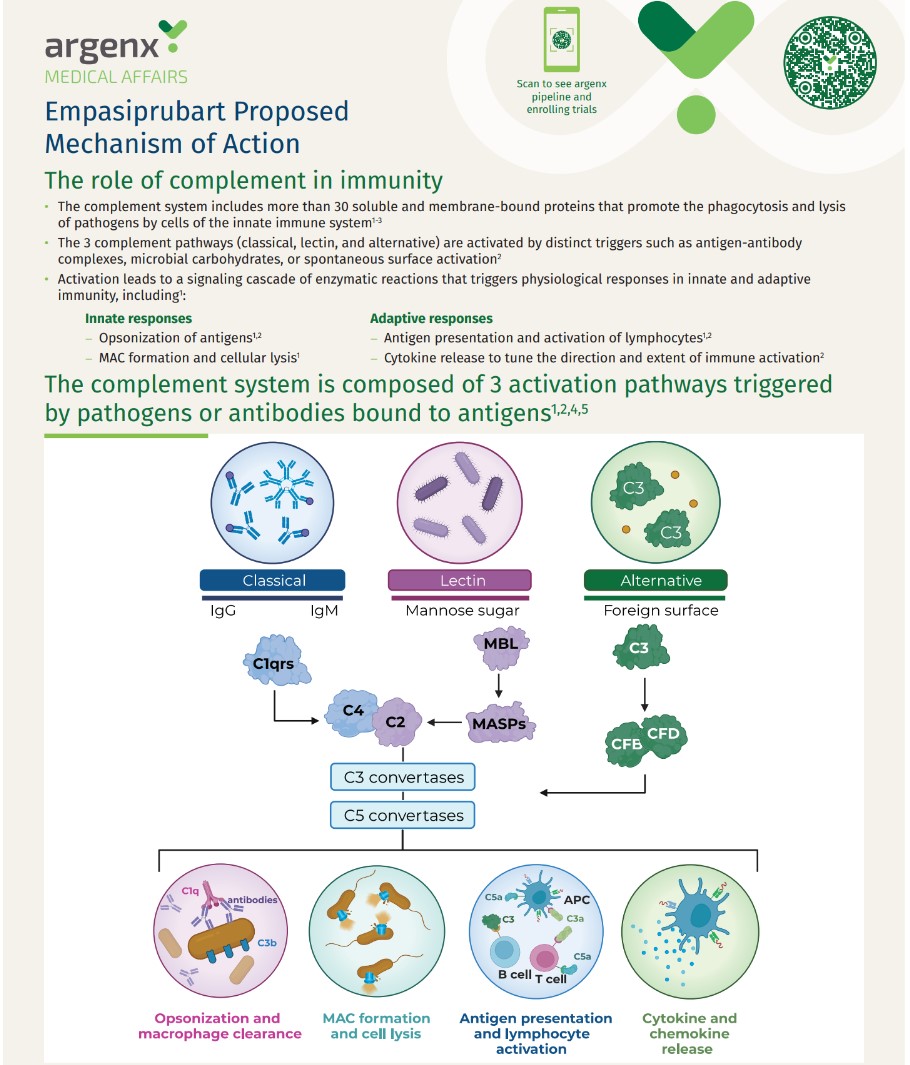
Empasiprubart 117 Proposed Mechanism of Action handout

MMN Disease State Handout
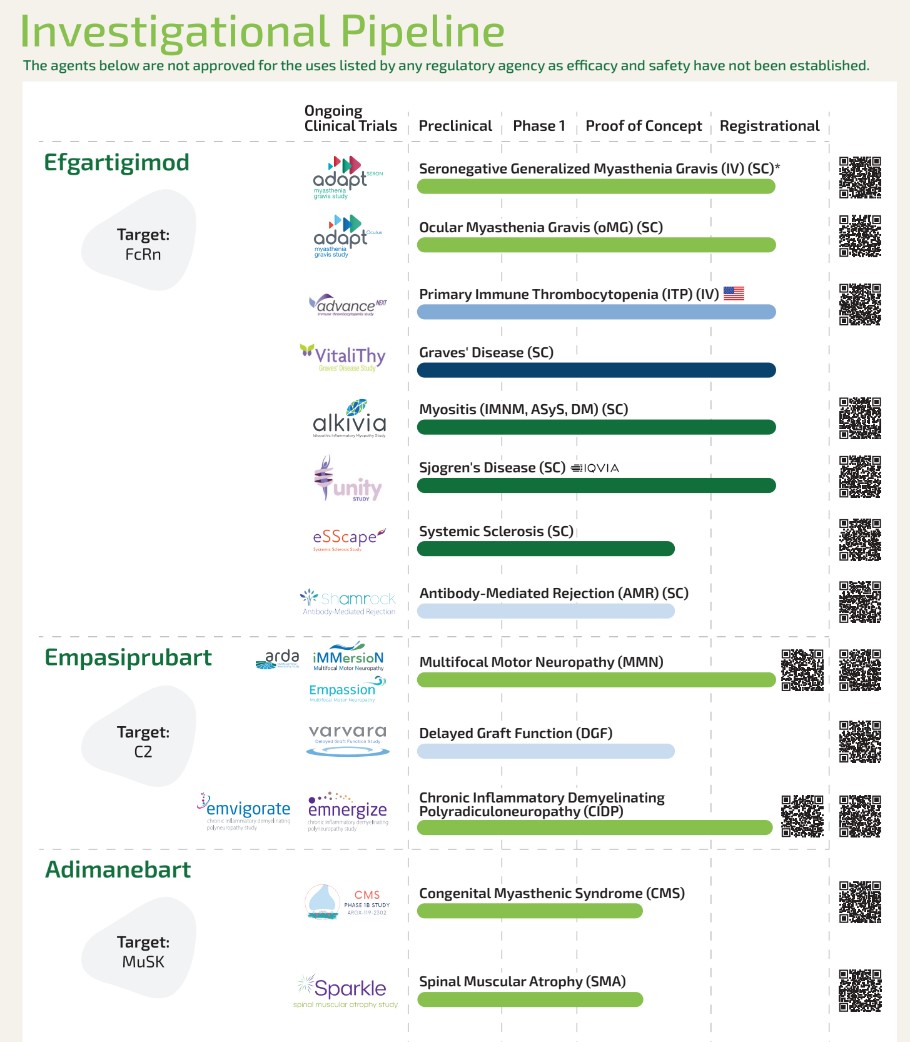
argenx Pipeline Handout for Congresses
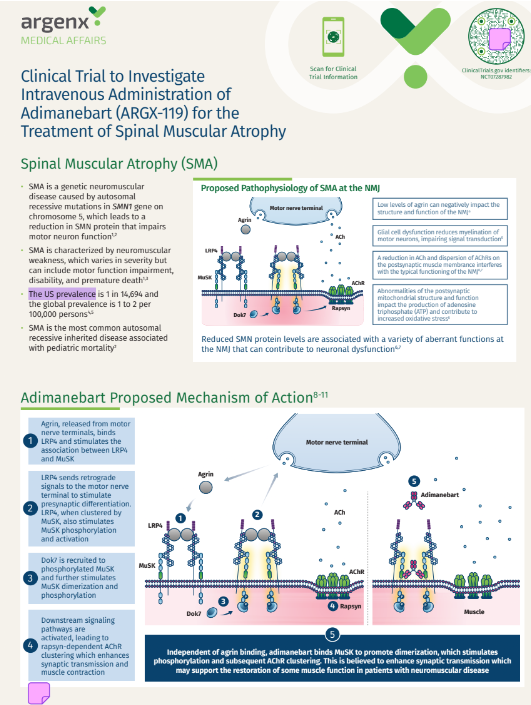
SMA Adimanebart (ARGX-119) Trial Awareness Handout

Adapt Jr Sc HCP Card

Vyvgart Pregnancy Study gMG CIDP HCP card

VYVGART + VYVGART Hytrulo Pregnancy Registry - Referring Physician Brochure
A U.S. brochure for prescribers and gMG‑treating HCPs introducing the ARGX‑113‑PAC‑2206 pregnancy post‑approval study

ADHERE REAL Study HCP Clinical Trial Quick Card

Emnergize HCP Card

Emvigorate HCP Card

Adapt Forward HCP Card

CMS natural history HCP Card

CMS Phase 1b HCP Card

Sparkle HCP Card

Adapt Early HCP Card

Adapt Jr HCP Card
Recent developments in the neuromuscular field.
Recent developments in the neuromuscular field.