To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Resource Library
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
To report SUSPECTED ADVERSE REACTIONS, contact argenx at 1-833-argx411 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Explore the mechanisms driving MMN and the clinical measures used to evaluate disease activity
Diverse autoantibodies targeting gangliosides have been observed in MMN.1
Antibodies of the IgG, IgM, and IgA classes have been detected in patients with MMN; common targets of these autoantibodies include gangliosides GM1 and GM2.1
Additional studies have identified autoantibodies targeting other neural antigens, including the mixture of GM1 with galactocerebroside or the disulfated heparin disaccharide NS6S.2
IgG
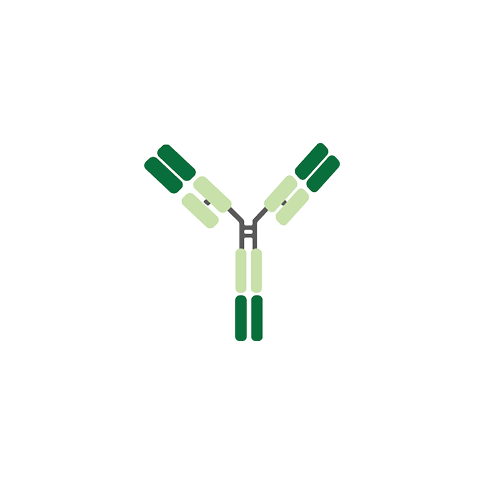
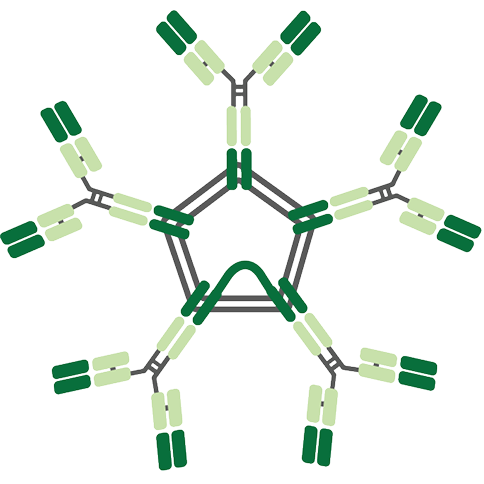
IgM
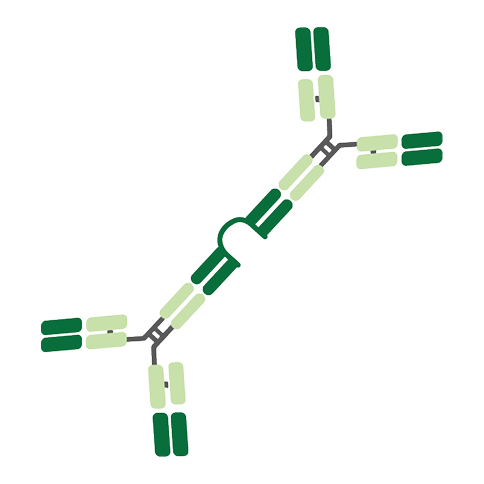
IgA
|
Class and target |
Percentage of patients with MMN |
|---|---|
|
Anti-GM1 IgM |
43% |
|
Anti-GM1 IgA |
5% |
|
Anti-GM1 IgG and IgA |
1% |
|
Anti-GD1b and anti-GM1 IgM |
9% |
|
Anti GM2 IgM |
6% |
|
Anti-GD1a, GalNAc-GD1a, GM1b, Gt1a, GQ1b, or SGPG IgM, IgA or IgG |
0% |
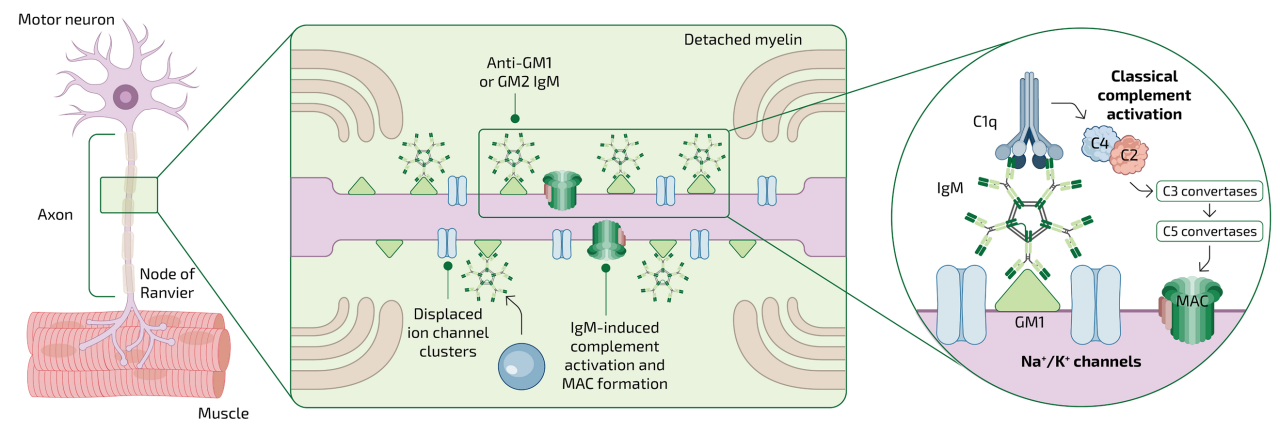
In MMN, IgM antibodies may cause nerve dysfunction by targeting ganglioside GM1, leading to conduction block through direct and complement-driven indirect mechanisms.11
Studies using patient sera suggested that IgM autoantibodies from patients with MMN can activate the complement cascade and that titers of anti-GM1 IgM antibodies correlate with classical complement pathway complex deposition.7,8
In these experiments, normal human sera served as a source of complement factors:
2015 in vitro analysis8
Vlam and colleagues found that anti-GM1 IgM antibodies from the serum of patients with MMN can activate complement and induce the deposition of complement activation products C3b and C5b-9 (the membrane attack complex) in vitro in a titer-dependent manner
2011 in vitro analysis7
Yuki and colleagues found a strong correlation between the deposition of complement factors C1q, C4b, and C3b in vitro and titers of anti-GM1 antibodies from the sera of patients with MMN
A study of patients with MMN demonstrated that complement activity correlates with MMN disease severity, as measured by muscle weakness and axonal loss.8,13
There are a number of tools available for the assessment of functional, patient-reported, or QoL outcomes in MMN:
Footnotes:
aData from a cross-sectional study of 88 patients in the Netherlands from January through December 2017; 81% had definite MMN, 18% probable, 1% possible; 73% were male; median age was 52 (27–78) years.1
Abbreviations:
C=complement factor; CAP-PRI=Chronic Acquired Polyneuropathy Patient-Reported Index; EQ-5D-5L=EuroQoL 5-Dimension, 5-Level; FSS=Fatigue Severity Scale; GalNAc=N-acetylgalactosamine; GD=disialoganglioside; GM=monosialoganglioside; GNDS=Guy’s Neurological Disability Scale; GQ=quadsialoganglioside; Gt=trisialoganglioside; 9-HPT=9 Hole Peg Test; HRPQ=Health Related Productivity Questionnaire; HRQOL=Health-Related Quality of Life; Ig=immunoglobulin; INCAT=Inflammatory Neuropathy Cause and Treatment; MAC=membrane attack complex; MMN=multifocal motor neuropathy; MMN-RODS=Rasch-built Overall Disability Scale for multifocal motor neuropathy; MRC=Medical Research Council; PGI-C=Patient Global Impression of Change; PGI-S=Patient Global Impression of Severity; PROM=Patient Reported Outcome Measure; QoL=quality of life; SGPG=sulfoglucuronyl paragloboside; TSQM=Treatment Satisfaction Questionnaire for Medication; VAS=Visual Analogue Scale.
References:
1. Cats EA, et al. Neurology. 2010;75(9):818-825. doi: 10.1212/WL.060133181f0738e; 2. Nobile-Orazio E, et al. J Neurol Neurosurg Psychiatry. 2014;85(7):754-758. doi:10.1136/jnnp-2013-305755; 3. Delmont E, et al. J Neuroimmunol.2015;278:159-161. doi:10.1016/j.jneuroim.2014.11.001; 4. Guo Z, et al. Int J Mol Sci. 2023;24(11x9558. doi:10.3390/ljms24119558; 5. Loser V, et al. Front Neurol. 2024;15:1495205. doi:10.3389/fneur.2024,1495205; 6. Krijgsman D, et al. Neurol Neuroimmunol Neuroinflamm. 2026;13(1):e200482. doi:10.1212/NXI.0000000000200482; 7. Yuki N, et al. J Neurol Neurosurg Psychiatry. 2011;82(1):87-91. doi:10.1136/jnnp.2010.205856; 8. Vlam L, et al. Neurol Neurommunol Neuroinflamm. 2015;2(4):e119. doi:10.1212/ NXI.0000000000000119; 9. Sathe A, Cusick JK. Biochemistry, Immunoglobulin M.[Updated 2022 Dec 19), In: StatPearls [Internet). Treasure Island (FL): StatPearls Publishing; Jan 2025. Available at: www.ncbi.nlm_.nih.gov/books/NBK555995/ (Accessed: November 2025); 10. Vlam L, et al. Nat Rev Neurol. 2011;8(1):48-58. doi:10.1038/nreurol.2011.175; 11. Harschnitz O, et al.J Clin Immunol. 2014;34(suppl1):S112-S119. doi:10.1007/s10875-014-0026-3; 12. Beadon K, et al. Curr Opin Neurol. 2018;31(5):559-564. doi:10.1097/WCO.0000000000000605; 13. Querol L et al. Nat Rev Neurol. 2017;13(9):533-547. dol:10.1038/nmeurol.2017.84; 14. Lee SY, et al. Ann Rehabil Med. 2021;45(3):167-169. dol:10.5535/arm.21106; 15. Neumann S, et al, In Vivo. 2017;31(5):917-924. doi: 10.21873/invivo.11147; 16. Kleyweg RP, et al. Muscle Nerve. 1991;14(11)1103-1109. doi:10.1002/mus.880141111; 17. Vanhoutte EK, et al. J Peripher Nerv Syst. 2015;20(3):296-305. doi:10.1111/jns.12141; 18. Vanhoutte EK. Peripheral neuropathy outcome measures standardisation(PeriNomS) study part 2: getting consensus. Doctoral Thesis. Maastricht University; 2015; 19. Moreno-Morente G, et al. Int J Environ Res Public Health. 2022; 19(16):10080. doi:10.3390/ijerph191610080; 20. Kumar RN, et al.J Occup Environ Med.2003;45(8):899-907. doi:10.1097/01.jom.0000083039.56116.79; 21. Atkinson MJ, et al. Health Qual Life Outcomes. 2004;2:12. doi:10.1186/1477-7525-2-12; 22. Liberato ACS, et al. Value Health Reg Issues. 2020;23:150-156. doi:10.1016/j.vhri.2020.07.578; 23. Krupp LB, et al. Arch Neurol. 1989;46(10)1121-1123. doi:10.1001/archneur.1989.00520460115022; 24. Shirley Ryan AbilityLab. Fatigue Severity Scale. Available at: www.sralab.org/sites/default/files/2017-06/sleep-Fatigue-Severity-Scale.pdf (Accessed: March 2026); 25. EUROQOL EQ-5D-5L user guide version 4.0. 2025. Available at: https://euroqol-domain.ams3.digitaloceanspaces.com/wp-content/uploads/2025/01/12124516/EQ-5D-5L-Userguide-2025-04.pdf (Accessed: March 2026); 26. Gwathmey KG, et al. Muscle Nerve. 2016;54(1)9-17. dol:10.1002/mus.24985; 27. Eremenco S, et al. Qual of Life Res.2022;31(12):3501-3512. dol:10.1007/s11136-022-03180-5; 28. Sharrack B, Hughes RAMult Scler.1999;5(4):223-233. doi:10.1177/135245859900500406; 29. Allen JA, et al. JAMA Neurol. 2020; 77(9):1159-1166; doi:10.1001/jamaneurol.2020.0781.